-
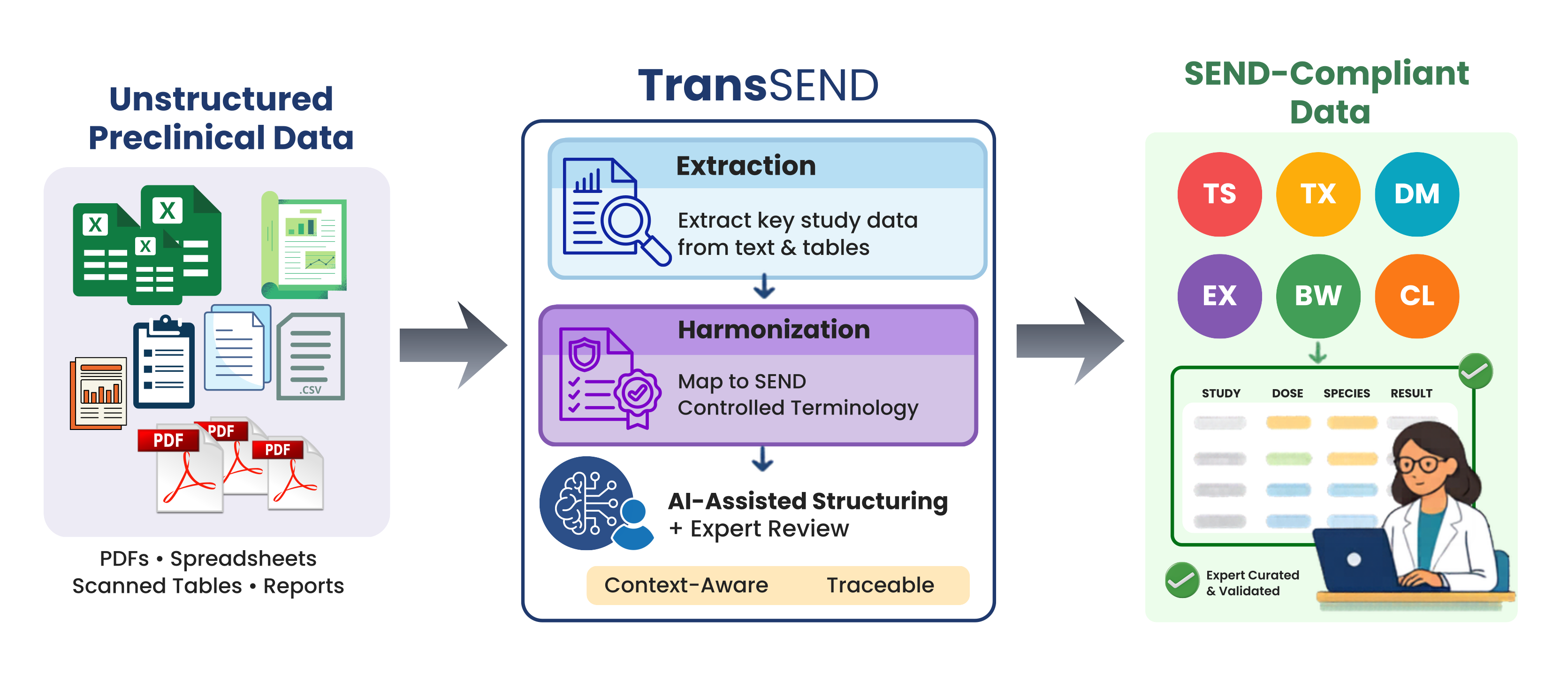
Making Preclinical Data Interoperable and Regulatory Aligned: The TransSEND Approach
Article March 23, 2026
Preclinical toxicology studies generate an enormous amount of information: dense tables, semi-structured datasets, and long-form reports hidden away in PDFs. Turning that mix into consistent, reusable, and regulatory-ready data is often slow, expensive, and frustrating. SEND (Standard for Exchange of Nonclinical Data) was created to tackle exactly this problem. Developed by the Clinical Data Interchange […]
-

MedBioinformatics Announces Participation in IHI JU VICT3R Initiative to Revolutionize Nonclinical Drug and Chemical Safety Evaluation with Virtual Controls
News September 16, 2024
[Barcelona, Spain, September 16, 2024] — MedBioinformatics today announced its involvement in the VICT3R initiative, a groundbreaking initiative aimed at transforming nonclinical drug and chemical safety evaluation. VICT3R is a public-private partnership supported by the Innovative Health Initiative Joint Undertaking (IHI JU), set to significantly reduce the number of animals used in experimental studies. The […]