-
Making Preclinical Data Interoperable and Regulatory Aligned: The TransSEND Approach
Article March 23, 2026
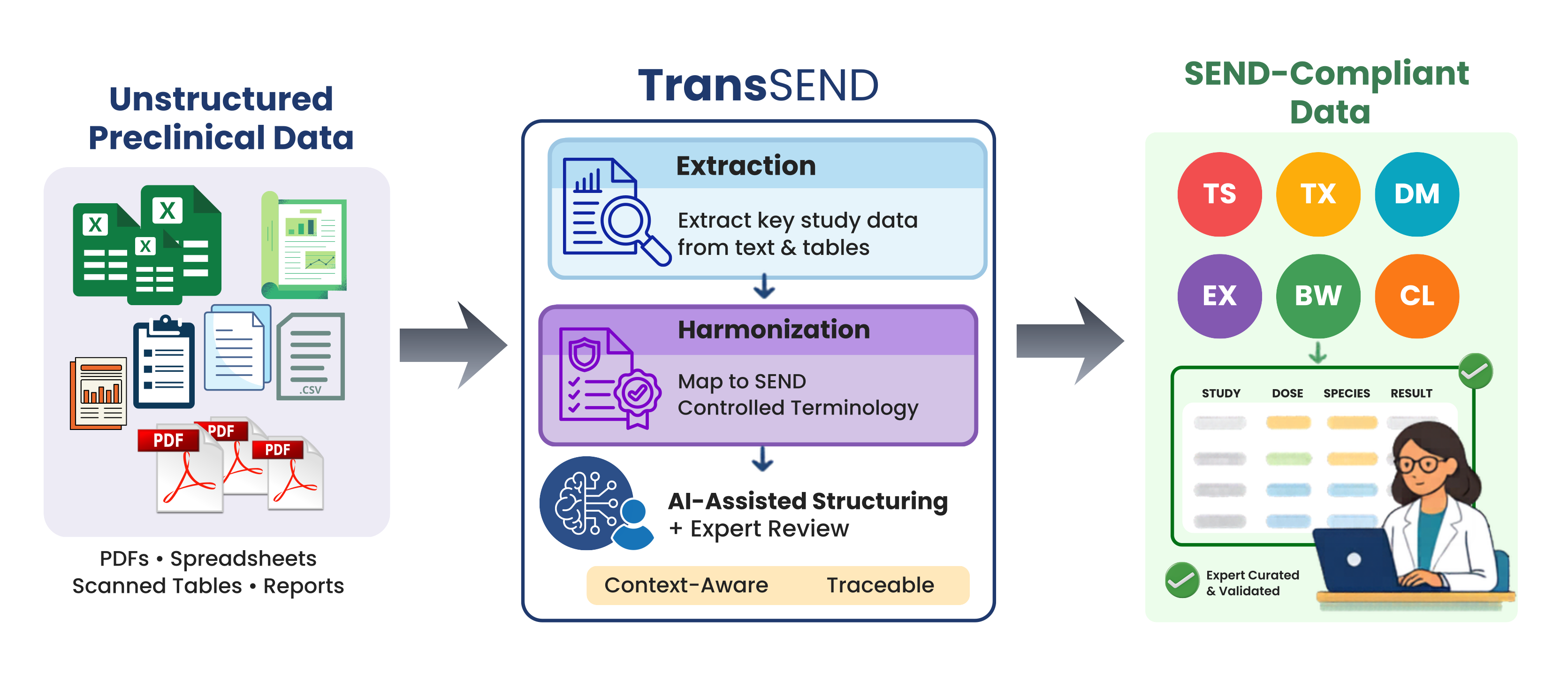
Preclinical toxicology studies generate an enormous amount of information: dense tables, semi-structured datasets, and long-form reports hidden away in PDFs. Turning that mix into consistent, reusable, and regulatory-ready data is often slow, expensive, and frustrating. SEND (Standard for Exchange of Nonclinical Data) was created to tackle exactly this problem. Developed by the Clinical Data Interchange […]